When you think about it, the connection between chemistry and magic has been running for a rather long time. Magic does not exist, obviously, at least in the sense we usually consider, but the Third Law of Clarke prevails: Any sufficiently advanced technology is indistinguishable from magic. Even better, as somebody said, any technology, no matter how primitive, is magic to those who do not understand it.
So when reactions behave unexpectedly under microwave conditions, we resort to magic (although it is not!). When we observe a disproportionate enhancement in the potency, selectivity or other properties after inserting a methyl group in a molecule, medicinal chemists call for a Magic Methyl effect.
As expected, it turns out that the effect is in fact caused by a series of physical phenomena, including favorable desolvation energetics, changes in metabolic stability (both decreases and increases), induced conformational effects and so on. So today, the Magic Methyl effect is sought in the same way the Philosopher’s Stone was coveted hundreds of years ago. When you already have an excellent molecule in hand, a further methyl insertion is always worth a try to see if magic happens. The introduction of new motifs into molecules at this final stage is appropriately known as Late-Stage Functionalization (LSF) so, what last-stage methylation reactions are available out there?
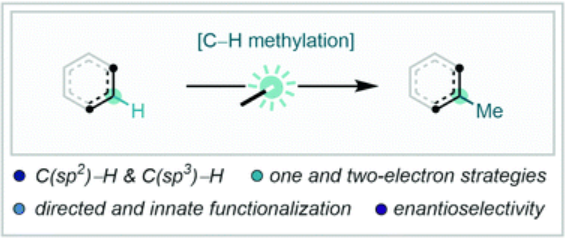
This review by Leitch and Dixon (University of Oxford, UK) covers different methods, including directed C(sp2)–H methylations, innate/direct C(sp2)–H methylations (we also recommend this excellent paper by Cernak et al. for a discussion about innate and guided LSF reactions), C(sp3)–H methylations, and enantioselective C–H methylations. As you will see, there are plenty of options available today.
Installing the “magic methyl” – C–H methylation in synthesis. Chem. Soc. Rev., 2021, XX, pp. XXX–XXX. See: https://doi.org/10.1039/D0CS00973C (Open Access)