Convergent Synthesis of Polysubstituted Aromatic Nitriles via Palladium-Catalyzed C–H Functionalization
Metal-mediated alkylations and cyanations are not new but, what about accomplishing both in the same step? A group in Canada shows how to prepare ortho alkyl and aryl nitriles in a one-pot reaction.
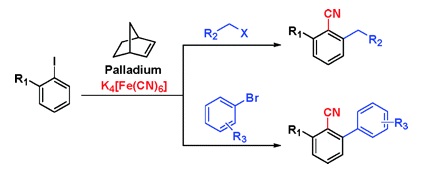
Tandem reactions are always interesting because of the obvious advantages of accomplishing several steps in a one-pot reaction. The research group of Lautens (Ontario, Canada) had published in 2006 a first report on the preparation of polycyclic benzonitriles. That work has now been extended to allow the preparation of nitriles with three different substituents using iodobenzenes as starting materials. The method uses a common palladium catalyst, norbornene and a metal cyanide to introduce the nitrile at the halogen position and a second group in ortho to that position. This second group can be an alkyl group, tethered or not, or even another a substituted aryl group. The reaction can thus yield ortho alkyl nitriles, ortho aryl nitriles, or new heterocycles. Those who contend with the preparation of small building blocks where the substituents are crowded on one side of the molecule will appreciate the beauty of introducing the two groups at the same time selectively.
J. Am. Chem. Soc., 2007, 129 (49), pp. 15372–15379. See: 10.1021/ja075599i