When you spend some time working in the field of Organic Synthesis you understand that Chemistry, as many other things, it is a matter of fashion. So far I have seen Solid Phase Organic Synthesis (SPOS), Combinatorial Chemistry, Catalytic Antibodies and others. Each trend has the corresponding group of advocates and critics, all moving around the fantastic benefits and results you could get from the new technology. So what else is new? Well, this time it is Flow Chemistry.
Flow Chemistry
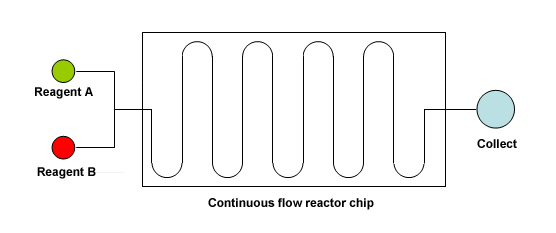
What is flow chemistry? Just a chemical reaction which is run in a continuously flowing stream instead of a ‘static’ volume contained in a vessel. To put it in the simplest way, you pump fluids containing reagents so they join into a mixer and then perform other operations (heating, cooling, etc.) so they react. If the reagents are reactive enough then mixing the two streams start the reaction. An important concept here is that molecules enter, react and leave the reaction system, thus avoiding protracted exposure to conditions that can promote the apparition of byproducts or impurities.
From the hardware point of view, any flow chemistry equipment uses one or two reservoirs of reagents, fine-quality pumps (flow control is critical), a reaction chip where mixing occurs and reaction takes place, and there is a system to collect the outcome. Many improvements can be done over this basic scheme (number of pumps, mixing chips, cooling/heating in different stages), etc…, so almost any reaction you can think of can be done in a flow chemistry reactor.
In fact, the success of flow chemistry in terms of reactivity arises from mixing. At the scale the flow reactors are operated, mixing becomes excellent and the heat exchange surface much more favorable than in a batch operation. Remember that doubling the size of the reaction multiplies the volume by 8, but the surface only by a factor of 4. That volume-surface ratio is specially critical for reactions at low temperature, where hot spots in the mixing interface are common, or in high temperature reactions where hot spots are located in the liquid nearest to the vessel’s wall. In that way microwave heating is similar to flow chemistry: temperature of the fluid being pumped through the system is homogeneous.
So to summarize, flow chemistry offers several important advantages over batch production: Reproducibility during the scale-up is higher because there are no problems due to the ratio volume:surface, which in turn reduces or even neglects the re-optimisation of chemistry at each stage; there is a precise control of mixing and reaction temperature; there is a geometric separation of starting material and products (i.e., once a molecule of product is formed it exits the reaction system); since flow chemistry reactors need only a fraction of the volume of batch operation, risk associated with hazardous intermediates is minimised; and finally, there is a rate acceleration effect due to superheating at elevated pressure.
All this advantages really make a difference. It has been demonstrated once and again that the time it takes to produce the first kilogram can be enormously reduced, and conditions can be easily transferred from microwave systems to flow chemistry systems (have I mentioned before that heating is quite similar between both systems?), thus avoiding costly modifications. There are now some companies selling flow chemistry reactors with a unit cost similar to the microwave reactors, papers are being published monthly and some top figures in chemistry are plugged to this new exciting field.
However, as with any other new exciting technology, things are not so wonderful. The biggest issue is that any reagent or material introduced into the system must be in solution, and that extends to any product being formed, so if precipitation of a product occurs, hey, you are in a trouble! This limitation extends to things like common catalysts. You simply cannot perform a coupling or hydrogenation reaction in the normal way: the catalysts, any catalyst, must be attached to a solid support so it can be used as filling in a column which is inserted in the flow system.
Some advocates of the flow chemistry are even trying to convince everybody that the future will be continuous: no more batch reactors. Well, that reminds me a of comment made by a professor eight years ago. The combichem fashion was still hitting hard, so this man told everybody that ‘in a near future any doctoral student will have in the bench a combichem robot to prepare hundreds of compounds, as today they have a rotavapor to eliminate solvents’. Yeah, sure. My own opinion is that flow chemistry will be another tool that will be used to solve some problems, but not all problems, as it has happened with many other new tools. But as usual, time will have the last word.