N-Alkylation of poor nucleophilic amine and sulfonamide derivatives with alcohols by a hydrogen autotransfer process catalyzed by copper(II) acetate
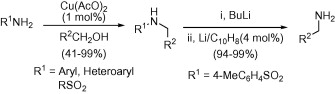
Alkylation of poor nucleophilic amines and sulfonamides with alcohols using a copper catalyst.
The selective alkylation of amines is a topic of great interest not only to medicinal chemists, but in many fields of chemistry. In the last months our own company has devoted some effort to develop and expand a new procedure to alkylate amines by using the Chan-Lam reaction. In the R&D section of this issue you can find more details on the later developments.
This paper by Yus (Alicante, Spain) is a complementary method for the chemistry being developed by us. Curiously, it uses also Cu(OAc)2 as catalyst, though in a completely different way. The source of the alkyl group here is an alcohol, through a process called hydrogen autotransfer, also known as borrowing-hydrogen. To make it simple, in a typical procedure the amine and an alcohol are mixed with the Cu(OAc)2 (10 mol%), tBuOK (100 mol%) in 1,4-dioxane at 130 °C for 2 days. The yields are usually excellent and the scope of the reaction quite good: anilines and heteroarylamines can be alkylated with many different benzylic alcohols. Interestingly this protocol can be applied also to the alkylation of sulfonamides.
One of the limitations of this method seems to be the time needed for the reaction: 2 days. It may be possible for microwaves to reduce that time. And I wonder if the tBuOK cannot be replaced with tBuONa.
Tetrahedron Letters 2010, 51 (2), pp. 325–327. See: 10.1016/j.tetlet.2009.11.009