Phenols, phenols, and phenols
Three papers using phenols as starting materials for organometallics couplings.
Phenols are ubiquitous products in nature. They are also common starting materials for a number of products, but working with unprotected phenols means being looking for trouble, and thus all work is usually done with the phenol in protected form but, what if the OH moiety is protected with a group that turns it into a very useful leaving group?
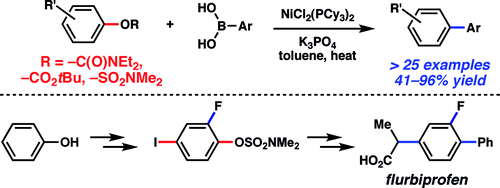
We present here three papers recently published on this topic. The first one is a work by Garg (UCLA, CA, USA). The paper describes the use of carbamates, carbonates, and sulfamates derived from phenols as useful substrates for nickel-mediated cross-coupling with boronic acids. Thus, carbonates and carbamates can be coupled using NiCl2(PCy3)2 (10 mol%), ArB(OH)2 (4 equiv) and K3PO4 (7 equiv) in toluene at 130 °C for 24 h. The yields obtained range from moderate to good, but at least the protocol opens the possibility of using other substrates when the corresponding aryl halide is not available. Very similar conditions are used with sulfamates but, in this case, the yields obtained are usually excellent, using a lower boronic acid ratio, something always interesting when you think about the price of some boronic acids. One example of a heterophenol (a hydroxyindole) is included. The utility of the sulfamate cross-coupling methodology is further demonstrated through the synthesis of flurbiprofen using orthogonal cross-couplings. A substrate containing an iodide and a sulfamate is selectively coupled using different nickel catalysts to obtain the desired product. As a final comment, the NiCl2(PCy3)2 is commercially available from Strem Chemicals at a very reasonable price, shows marked stability toward air and water, and can be used on the benchtop rather than in a glovebox.
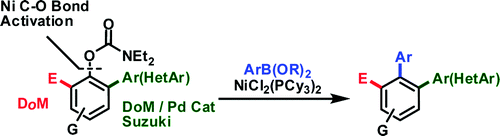
The second paper by Snieckus et al. (Queen’s University, Ontario, Canada) is closely related to the work by Garg’s group with carbamates. In this case Snieckus is using the carbamate as directing group for an ortho metallation but, since it is a potential leaving group for an organometallic coupling, they started trying the conditions reported previously by Garg and came up with the same solution: NiCl2(PCy3)2. However, the protocol is slightly different. In a typical example, once the DoM work is done and the ortho substituent is in place, the carbamate is reacted with a boroxine, NiCl2(PCy3)2 (5 mol%), PCy3HBF4 as the ligand (10 mol%), and K3PO4 as the base in o-xylene at 150 °C for 5–20 h. The yields obtained go from 20–30% up to quantitative, but they are applied with excellent results to some nitrogen heterocycles, specially 3-pyridines.
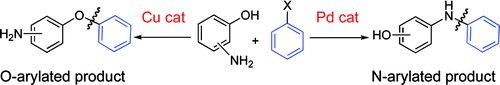
The third paper by Buchwald et al. (MIT, Mass., USA) expands the options for couplings with aminophenols, developing orthogonal protocols for selective N- or O-coupling. Thus, for 3-aminophenols, two sets of conditions are described for the O-arylation with iodides and bromides using CuI as the catalyst and another two sets of conditions are described for the N-arylation with bromides and chlorides using BrettPhos precatalyst as the palladium source. Interestingly, although the O-arylation results are better using the iodides, for the N-arylation, the use of chlorides or bromides is equivalent. 4-Aminophenols are also tried, with O- and N-arylation conditions being described with CuI and BrettPhos precatalyst, respectively. Finally, 2-aminophenol is explored, but here only a selective N-arylation protocol is described due to the ability of the substrate to form a five-membered chelate. These conditions will be useful for those medicinal chemists attempting selective arylations with substrates like these.
Suzuki–Miyaura Coupling of Aryl Carbamates, Carbonates, and Sulfamates. J. Am. Chem. Soc., 2009, 131 (49), pp 17748–17749. See: 10.1021/ja906477r
Orthogonal Cu- and Pd-Based Catalyst Systems for the O– and N-Arylation of Aminophenols. J. Am. Chem. Soc., 2009, 131 (47), pp 17423–17429. See: 10.1021/ja9081815
N,N-Diethyl O-Carbamate: Directed Metalation Group and Orthogonal Suzuki–Miyaura Cross-Coupling Partner. J. Am. Chem. Soc., 2009, 131 (49), pp 17750–17752. See: 10.1021/ja907700e