PyFluor: A Low-Cost, Stable, and Selective Deoxyfluorination Reagent
A new reagent for the replacement of aliphatic hydroxy groups by fluorine.
A new reagent for the replacement of aliphatic hydroxy groups by fluorine.
In previous entries of this blog we have commented new papers about new fluorination reagents. In the last year there has been a steady stream of novelties on this field, and this paper by Doyle et al. (Princeton University, NJ, USA) is another addition to the toolbox.
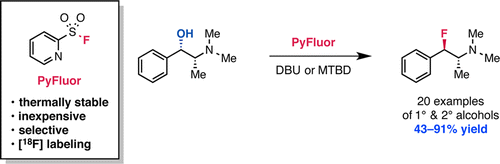
PyFluor (2-pyridinesulfonylfluoride) is a low cost, thermally stable and easy to prepare reagent that can transform aliphatic hydroxy groups into fluorides. The transformation is done by stirring the alcohol with PyFluor and a base (DBU or MTBD) in toluene at rt for a couple of days. In the more than 20 examples included, the reaction works nicely with a wide pool of substrates, including sugars, steroids, benzylic alcohols and secondary alcohols. In terms of diastereoselectivity, the results obtained are good: no epimerization, just inversion. It has also good regioselectivity and unlike other reagents like DAST the elimination problem is uncommon. The multigram synthesis of PyFluor is described in the paper. So what are you waiting for?
J. Am. Chem. Soc. 2015, 137 (30), pp 9571–9574.
See: 10.1021/jacs.5b06307