Biochemical and structural characterization of a novel thermophilic esterase EstD11 provide catalytic insights for the HSL family
A few years back, we participated in the research project HOTDROPS, whose goal was to search for new biocatalysts through the development of an ultrahigh-throughput platform. The project thus combined three sciences: enzymology, microfluid technology, and organic chemistry.
We are happy to present you now the latest article on some of the work carried out within that project: the identification and characterization of a new esterase enzyme that was found to show remarkable thermal stability and performance with substrates of industrial interest.
Esterases and lipases have a great number of industrial applications as they accept a broad range of non-natural substrates. Many of them show high stability in organic solvents, and even good regio- and stereoselectivity. Frequently they also show “catalytic promiscuity”, that is, the ability to catalyze more than one type of chemical reaction.
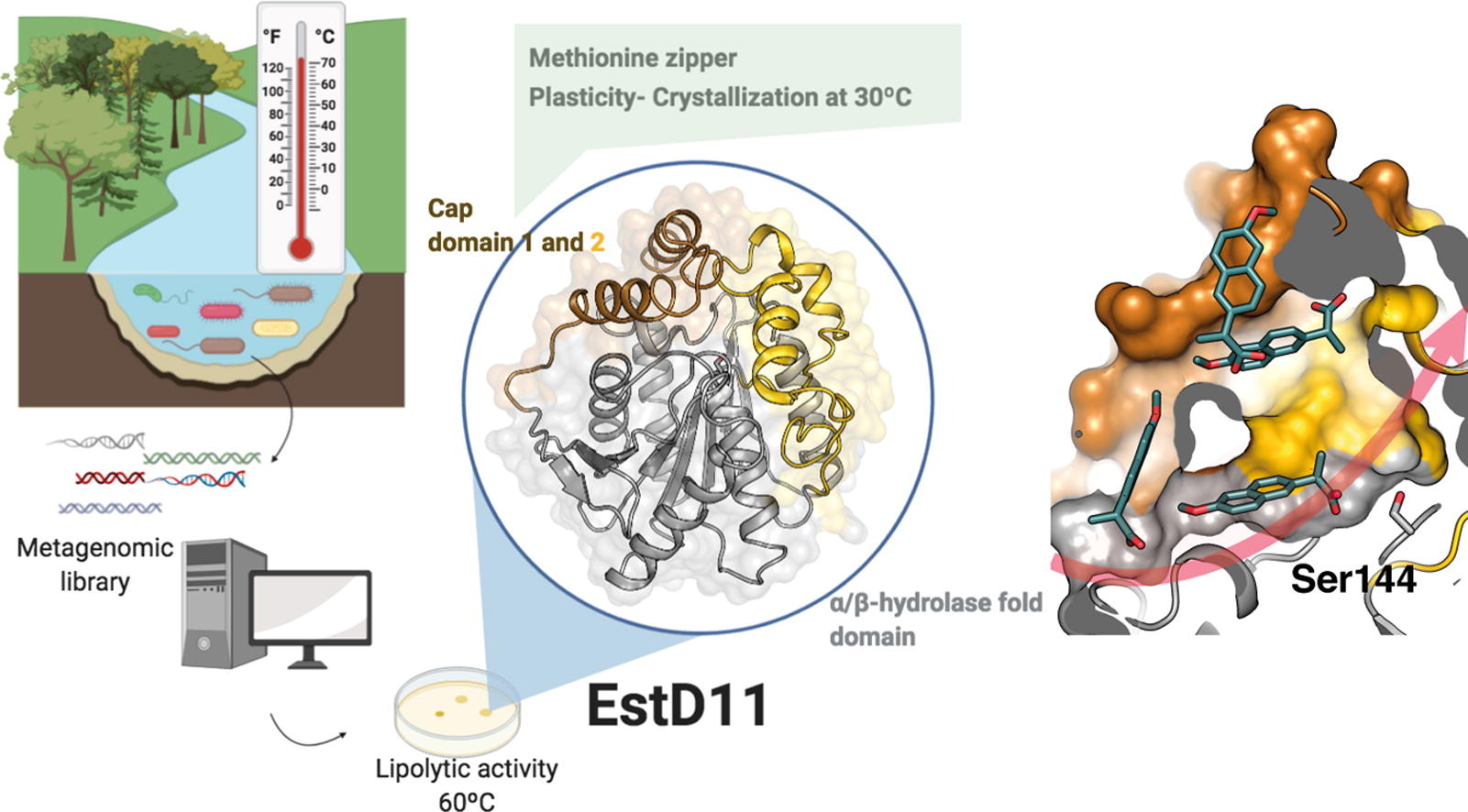
Enzyme EstD11 was extracted from a hot spring in Galicia (north-west of Spain and the home of GalChimia HQ) and is a thermophilic and thermostable esterase with an optimum temperature of 60°C.
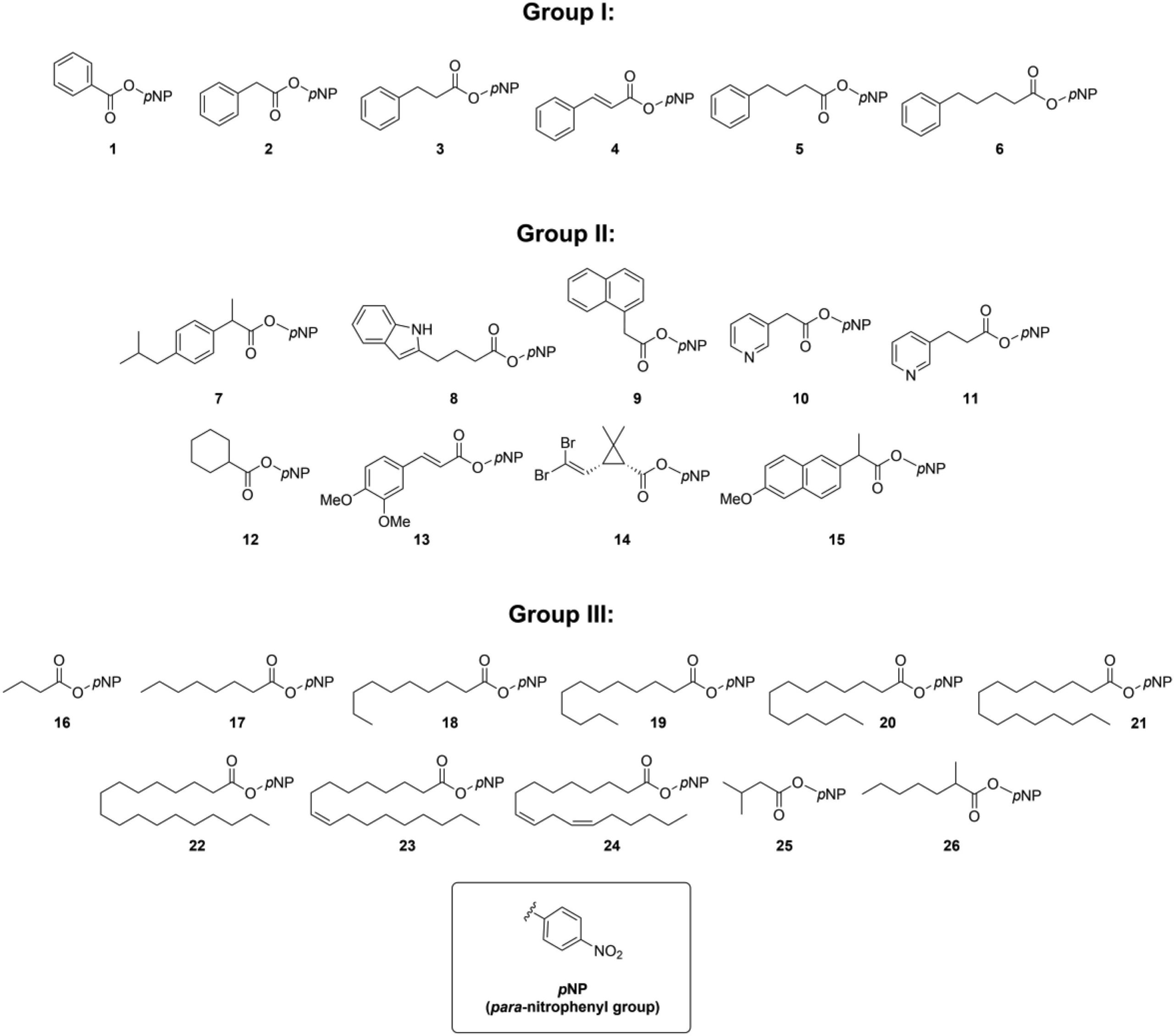
A detailed analysis of its activity was performed at GalChimia, whereby a wide variety of ester substrates was synthesized to assess the substrate scope of the new enzyme. A total of 26 esters were prepared with varied chain length, steric hindrance, aromaticity and flexibility of the linker between the carboxyl and the alcohol moiety of the ester. While the best substrate was pNP cyclohexanecarboxylate, EstD11 showed significant activity against 16 substrates, indicative of the substrate promiscuity of this enzyme.
The paper also presents the detailed characterization of the structure of this protein that accounts for the observed catalytic activity.
The HOTDROPS project ran from 2013 to 2017 with funding from the European Union through the Seventh Framework Programme – Industry-Academia Partnerships and Pathways – Marie Curie Actions (FP7-IAPP-Marie Curie).