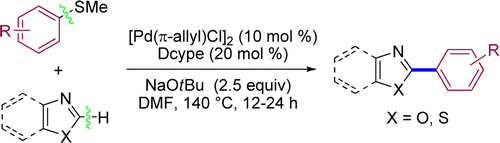Palladium-Catalyzed Coupling of Azoles or Thiazoles with Aryl Thioethers via C–H/C–S Activation
Coupling of azoles and thiazoles with aryl thioeters when nothing else is available.
Another example of C-H activation and we-can-couple-everything. The group of Wang et al. (University of Science and Technology of China) has published a curious paper. They activate the C-H bond in azoles and thiazoles and couple it with aryl thioethers, not less. The catalytic system is [Pd(p-allyl)Cl]2-Dcype (don’t worry, it is commercial) and NaOtBu as base. The pool of examples is, as usual, restricted. One example with pyridine is included with a 84% yield, so it could be potentially useful with other Het-SMe substrates around. They demonstrate also that aryl methiltioethers are not the only ones working; other S-things work. Could be useful when you have no other coupling partners available, and you are working with azoles.
Org. Lett. 2015, Article ASAP.
See: 10.1021/acs.orglett.5b00510