Easily Attachable and Detachable ortho-Directing Agent for Arylboronic Acids in Ruthenium-Catalyzed Aromatic C–H Silylation
Introduction of diversity in available boronic acids.
The more we have, the more we want. There currently is a huge number of boronic acids available from commercial suppliers, but since the preparation of boronic acids can be troublesome, time consuming and costly, very often medicinal chemists must adapt the exploration of the chemical space to those structures available. What if you have a method to prepare more than one substrate using just one boronic acid?
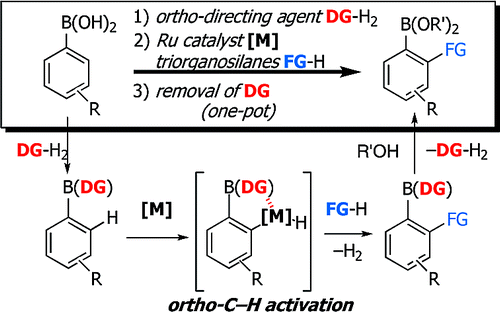
This paper by Sugimone (Kyoto University, Japan) can give some ideas. The starting point is that instead of preparing the desired boronic acid from scratch, functionalization of easily available organoboronic acids is better. The idea is tested on a ruthenium-catalyzed C–H silylation reaction. For this activation to work, an activating group must be reacted with the boronic acid so that an ortho-directing group is available. 2-Pyrazol-5-ylaniline (pza) was chosen as the activating group. The reaction of a boronic acid with this product leads to an intermediate protected boron compound, which is in turn subjected to the silylation. Deprotection of the group pza allows the Suzuki coupling to proceed. Finally, standard protocols (like the Tamao–Fleming oxidation) can be used to transform the alkylsilyl group into other moieties. Additionally, 2-pyrazol-5-ylaniline must be prepared in a three-step sequence.
Do not tell me: too lengthy. Although the method still needs some refinements, there are some points to be considered. First, the protection-silylation sequence can be done in a one-pot procedure. Second, the deprotection can be substituted by the transformation into a pinacol ester, which can be more efficiently isolated and can also be used for the Suzuki coupling. If the acidic deprotection is chosen instead, the pza can be recovered. Maybe, it is still at an early stage, but is nevertheless promising.
J. Am. Chem. Soc., 2009, 131 (22), pp 7502–7503. See: 10.1021/ja902314v