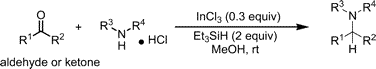Highly Chemoselective Reductive Amination of Carbonyl Compounds Promoted by InCl3/Et3SiH/MeOH System
A new procedure for reductive amination in protic solvents.
As we have commented in previous issues of this newsletter, reductive amination is one of the most important reactions in chemistry. Probably the most cited paper on this issue is the paper of Abdel Magid (J. Org. Chem. 1996, 61, 3849) describing different conditions, solvents and reducing agents to carry out the reaction. The common conditions described in the paper involve the use of sodium triacetoxyborohydride as an alternative to cyanoborohydride, which is toxic. Although other reductors can be used, it seems medicinal chemists have stuck to borohydrides.
The group of Yang (Hong Kong, China) describes now a new reducing system which is claimed to be more chemoselective and ecofriendly. The reductor used is Et3SiH, a hydride previously used in combination with several additives like TFA, TiCl4, Bu2SnCl2 and others. However, most of this additives have one problem or another: water sensitivity, low chemoselectivity or substrate dependence. The introduction of InCl3 overcomes all these problems allowing the reaction to be performed in protic solvents with good results.
The typical procedure involves adding InCl3 (0.3 eq) and Et3SiH (2 eq) to a solution of the carbonyl compound and the amine hydrochloride in MeOH at room temperature. A pool of amines with sensitive groups and carbonyl compounds (ketones and aldehydes) were evaluated, with yields ranging from very good to excellent.
J. Org. Chem., ASAP Article. See: 10.1021/jo8016082