Umpolung Amination: Nickel-Catalyzed Coupling Reactions of N,N-Dialkyl-N-chloroamines with Diorganozinc Reagents
An inverted alternative to the usual Buchwald–Hartwig amination.
There are two widely spread reactions for the coupling of amines with (hetero)aryl halides. The first one is the Buchwald–Hartwig reaction, a palladium-catalyzed coupling of an amine with a halide partner. The second one is the Ullmann coupling, an older copper-catalyzed version of the same coupling that has been rediscovered, improved, and expanded in the last decade. The Chan–Lam reaction is related to the Ullmann one, where the coupling partner is a boronic acid instead of the halide. All these reactions have one point in common: the amine is always one of the partners.
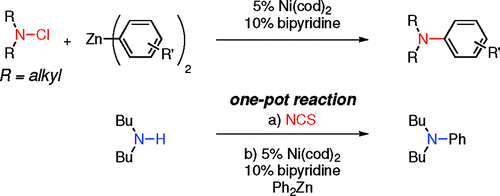
The work by Jarvo et al. (Irvine, CA, USA) introduces the umpolung concept in this arena. Thus, the nucleophilic amine is turned into the electrophilic component of the reaction by generation of the corresponding N-chloroamine with NCS and the coupling partner is now a diorganozinc reagent. The initial results are quite good and the coupling of a pool of N,N-dialkyl-N-chloroamines with Ph2Zn gives the products in good yield. Other diorganozinc reagents can be used with similar results. To finish, a one-pot procedure is presented, where the amine is oxidized by NCS and Ni(cod)2 (5 mol%), bipyridine (10 mol%), and the organozinc (only Ph2Zn is tested) are mixed in DMA/THF at 0 °C for 4.5 h to obtain the desired product.
J. Am. Chem. Soc., 2009, 131 (43), pp 15598–15599. See: 10.1021/ja907038b