A New Efficient Synthesis of Isothiocyanates from Amines Using di-tert-Butyl Dicarbonate
The preparation of many sulfur-containing heterocycles relies on the availability of isothiocyanates. See how to prepare them better.
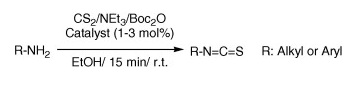
Isothiocyanates are chemoselective electrophiles in bioconjugate chemistry because of their tolerance toward aqueous media. They are also key intermediates in the synthesis of sulfur-containing heterocycles. Although known methods are efficient, Boas et al. (Copenhaguen, Denmark) have developed an even more efficient method that yields just the isothiocyanate… and volatiles.
As expected, the method uses as substrates amines, which are first treated with CS2 and Et3N and subsequently with Boc2O. The intermediate dithiocarbamate reacts with di-tert-butyl dicarbonate to give the isothiocyanate, COS, CO2, and t-BuOH. The authors found that a catalytic amount of DMAP or DABCO (1–3 mol%) increased the reaction rate significantly, with visible evolution of gas from the reaction mixture. Since all the remaining reagents are also volatile, simple evaporation of the crude leaves a high purity product in almost quantitative yield. A pity we cannot do the same with other reactions…
Tetrahedron Lett., 2008, 49 (19), pp 3117–3119. See: 10.1016/j.tetlet.2008.03.045